 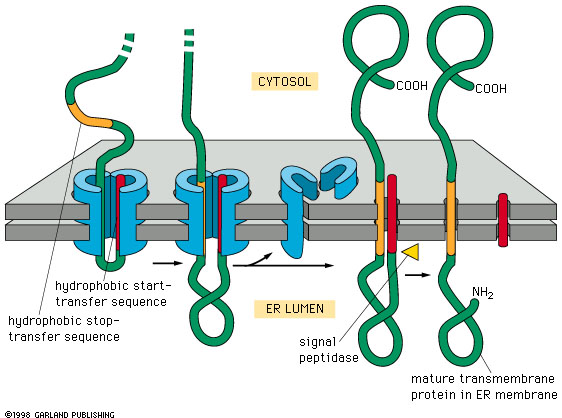 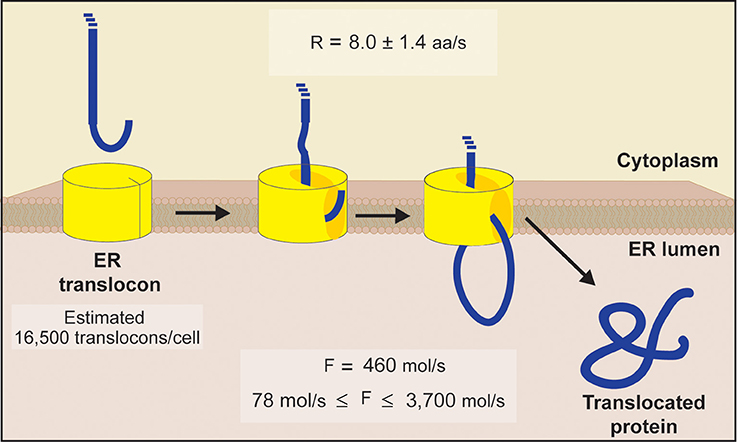
Both soluble luminal proteins and integral membrane proteins are folded in the ER and undergo quality control before being released into the secretory pathway. The endoplasmic reticulum (ER) represents the organelle with the largest flux of proteins, with over 40% of proteins translocated into the ER before trafficking to other organelles or secretion from the cell. Targeting proteins for degradation from within organelles requires a detailed understanding of local organellar protein quality control systems. However, many proteins that originate from the lumen of membrane-encapsulated organelles are also degraded using the cytosolic proteasome and degrons for these organelles are mysterious 12. All known degrons target cytosolic proteins for ubiquitination and degradation by the proteasome and the identification of degrons for a few key ubiquitin ligases has enabled exploitation of the proteasome to facilitate targeted protein degradation of “undruggable” cytosolic proteins 11. Even with a wide range of physiological roles for protein degradation, degrons are still unidentified for most E3 ubiquitin ligases. Recent systems-level analyses have expanded the availability of known degrons broadly 6, 7, with a series of “C-end rules” 8, 9, and additional variations of the N-degron pathways (reviewed in 10). The first degrons to be discovered were at the amino-terminus of proteins 3 and these N-degrons were eventually summarized as the “N-end rule” 4, and later the “N-degron pathways” 5. Inherent degrons can be shielded when proteins are appropriately-folded or incorporated into larger protein complexes. Inherent degrons are features of the primary polypeptide sequence formed by linear or conformational epitopes. Acquired degrons are post-translational modifications that can be based on proteolytic cleavage, phosphorylation, or acetylation. The specificity of the ubiquitination process is primarily driven by the E3 ubiquitin ligases and a major challenge in ubiquitin biology is the identification of the sequences that target proteins for degradation (called “degrons”).ĭegrons are short (usually <10 amino acids), linear motifs and, by definition, are sufficient to confer degradation when transferred to otherwise stable proteins. 
cerevisiae there is one E1, eleven E2s, and >60 E3s mammals have two E1s, ∼40 E2s, and >600 E3s 2. Ubiquitination requires the concerted action of the ubiquitination cascade comprising E1 ubiquitin activating enzymes, E2 ubiquitin conjugating enzymes, and E3 ubiquitin ligases. The ubiquitin proteasome system is the primary cellular degradation route, accounting for over 80% of protein degradation 1. Protein degradation plays an essential role in regulating diverse cellular processes including cellular signaling, metabolic adaptation, and cell cycle regulation. Future work is required to get a more detailed view of the properties of this degron, the mechanisms underlying its recognition by ER-resident and cytoplasmic factors, and the in vivo relevance of the findings. The authors provide solid evidence that this sequence functions as a "degron" for ER proteins. This important study identifies a short amino acid sequence that, when fused in multimeric form to the amino termini of luminal ER proteins, initiates proteasomal degradation via the Hrd1 ER quality control ubiquitin ligase complex. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
